Life Began with RNA Theory Gets Renewed Support
New research published in Nature by German scientists revealed that RNA molecules can grow peptides without ribosomes. It supports the RNA world hypothesis, which proposes that the first organisms were based on RNA strands, a molecule that can act as storage for genetic information and as catalysts for chemical reactions.
The RNA world concept first proposed by Alexander Rich in 1962 serves as a fundamental pillar of life’s origin theory, it hypothesizes that life evolved from increasingly intricate self-replicating RNA molecules. But the question of how RNA-based life evolved to the modern stage, in which proteins became catalysts and RNAs became an information carrier, is one big dilemma. This publication provides a new way of thinking about how peptides were built.
On modern Earth, “canonical” RNA bases – adenine (A), cytosine (C), guanine (G), and uracil (U) make up the genetic letters in messenger RNAs (mRNAs) that the ribosomes translate into proteins. Moreover, modified versions of the standard bases, “non-canonical” bases, are also ubiquitous in modern cells’ ribosomes, carrying out a variety of roles, eg. stabilizing binding of canonical RNAs and transfer RNAs (tRNAs) that help ribosomes translate genetic code into proteins.
Related article: The 6 Companies Leading the Resurgence in RNA Editing R&D
Four Canonical RNA Bases: A, C, G & U
Lead researcher Felix Müller, Luis Escobar and colleagues analyzed the chemical properties of non-canonical nucleosides to understand initial processes that may have led to the emergence of an RNA-peptide world, then modern life. They demonstrated that some non-canonical bases could bind the building blocks of proteins – amino acids, indicating these bases could also link peptides together into more complex peptides.
Beginning with pairs of complementary RNA strands, one “donor” and the other “acceptor”, the team added non-canonical base m6aa6A to the donor strand and mnm5U to the acceptor strand. When the complementary donor and acceptor strands are bound together, the mnm5U grabs ahold of the amino acid on m6aa6A, and with addition of energy (heat), m6aa6A releases and passes its amino acid over to mnm5U, and the complementary strands are disassociated and drifted apart.
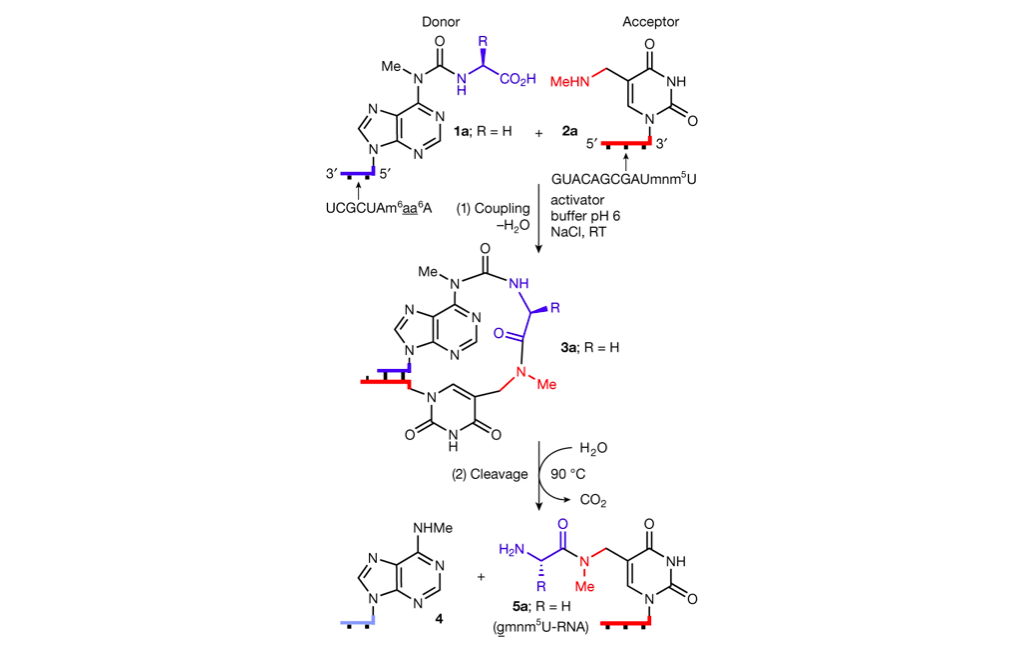
Considering early ribonucleic acid machinery’s low efficiency, longer peptides can be generated by coupling peptide fragments with these donor strands containing a longer peptide, suggesting the possibility to produce more complicated molecules with few reaction steps.
The team expressed that RNAs has the potential to create peptide self-decorating RNAs and hence an RNA-peptide world. The formed molecules are comparatively stable, and it is believed that, at some point, evolved and advanced to activate amino acids by adenylation, then transfer them onto the ribose OH groups to compartmentalize reactions in large and hydrophobic compartments, essential in launching the RNA-peptide world to the symbolic ribosome-focussed translational process we familiar with today.
Related article: TALED Tool Launches a New Era of Mitochondrial Genome Editing
©www.geneonline.com All rights reserved. Collaborate with us: service@geneonlineasia.com








