U.S.-German Joint Discovery May Spark Breakthrough in Diabetes Research
Researchers from the U.S.-based Van Andel Institute and Max Planck Institute of Immunobiology and Epigenetics in Germany have identified two distinct subtypes of insulin-producing beta cells (ß cells), each possessing distinctive characteristics. The findings of this study might contribute to a better understanding of and treatment for type 1 and 2 diabetes.
Led by Dr. John Andrew Pospisilik, Professor of the Department of Epigenetics at Van Andel Institute, the U.S.-German team reported their findings in an article published in the March 21 issue of the journal Cell Metabolism.
Related article: CDC Says Childhood Diabetes Could Increase Nearly 700% By 2060
ß Cells Play a Critical Role in the Pathogenesis of Diabetes
β cells are unique cells found in the pancreatic islets that are responsible for the synthesis, storage, and secretion of insulin, regulating blood sugar levels by designating dietary sugar for immediate use or storage. Hence, these cells are important for the regulation of metabolic processes and their loss or functional impairment can contribute to the pathogenesis of both type 1 and type 2 diabetes.
In Type 1 diabetes, the destruction of β cells by autoimmune attacks renders them unable to produce insulin, compromising the body’s ability to respond to blood glucose levels. Type 2 diabetes arises from insulin resistance due to genetic or dietary factors. This phenomenon can make β cells become overworked and exhausted from being overstimulated, leading to a significant reduction in both cell volume and function.
Discovery of Two New Subtypes of β Cells
β cells were once generally regarded as a single, homogenous cell population. Previous research demonstrated that these cells can actually be subdivided into at least four subtypes depending on their differential expression of certain cell surface proteins, and each subtype has a different gene expression profile and secretes insulin in a different manner. Besides, it was found that the distributions of β cell subtypes could be altered in the majority of patients with type 2 diabetes.
This recent study is particularly significant in that it is the first to explicitly describe specific ß cell subtypes. These findings have been confirmed in both mouse models and human ß cell samples.
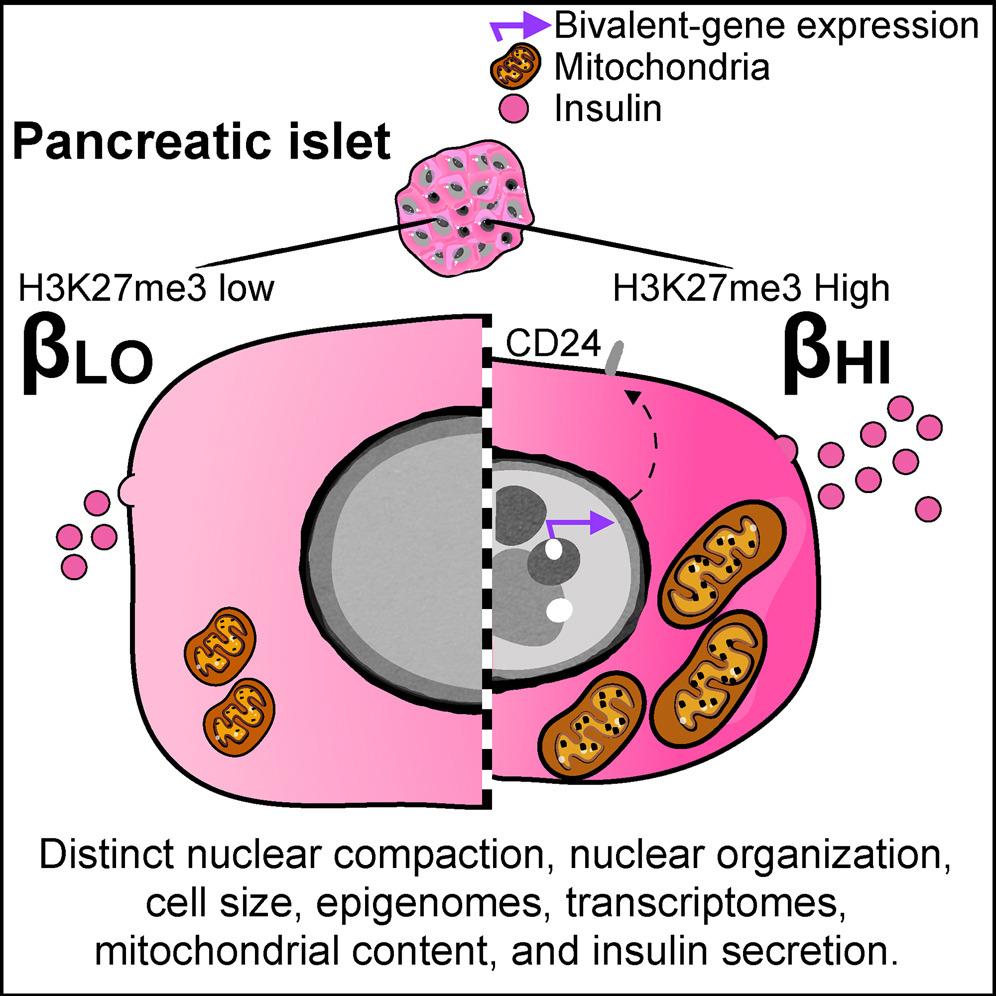
The two subtypes identified in this study, which the authors described as ßHI and ßLO, differ in many ways such as specific cell functions, size, shape, cytosolic and nuclear ultrastructure and epigenomic features. They also exhibit contrasting patterns of surface markers, which help cells send and receive chemical messages. Similar to previous studies, researchers observed a difference in the distribution of β cell subtypes in type 2 diabetes, in which ßHI cells appeared to be present in a higher ratio than ßLO cells.
Distinction Between Subtypes Results From Histone Mark Heterogeneity
The paper also illustrates different ways in which the two β cell subtypes can be distinguished. On the one hand, the subtypes can be separated by the presence or absence of a protein called CD24, which acts as a marker that allows the targeting of ßHI cells and not ßLO cells.
On the other hand, the team identified that the two β cell subtypes are epigenetically distinct, as they found that βHI cells exhibit roughly 4-fold higher levels of H3K27me3, an epigenetic modification to the DNA packaging protein Histone H3, showing that amount of specific epigenetic mark can be a decisive factor contributing to the differentiation into βHI or βLO cells.
Promising Discovery That Opens New Door to Diabetes Research
The new findings from the U.S.-German team suggest several potential paths that could inspire future research on diabetes treatments. According to Prof. Pospisilik, understanding these two subtypes of β cells and their relationship to each other may provide a clearer picture of diabetes and offer new opportunities for treatment.
For instance, the distinction between the presence and absence of CD24 cell surface markers may provide hints for the development of more precise diabetes treatment strategies and tools for in-depth investigation of each cell type.
Further, the team’s discovery of a difference between the two subtypes in terms of epigenetic modification is rather groundbreaking because the mechanism relies solely on epigenetics without involving transcription factors. This may offer an unprecedented target for potential new treatments for diabetes. “The key here is that epigenetic changes can be reversed, which opens a whole host of questions with implications for treatment,” said Prof. Pospisilik.
©www.geneonline.com All rights reserved. Collaborate with us: service@geneonlineasia.com








