Is Bayer’s Entry to the Gene Therapy Market Worth it’s Investment?
By Ruchi Jhonsa, Ph.D.
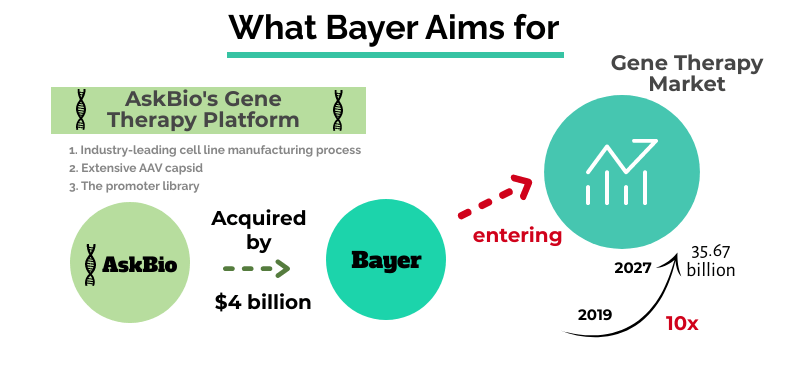
Bayer AG, the German drug pharma, is purchasing US-based gene therapy pioneer, Asklepios BioPharmaceutical, AskBio for up to $4 billion. Marking the biggest pharma deal of the company since 2006, the acquisition is Bayer’s move to enter the booming field of gene therapy, which according to the experts, is projected to reach USD35.67 billion by 2027- 10 fold higher than the 2019 market size.
Seeds of AskBio were planted by Jude Samulski, a pioneer of AAV vectors, in 2001. The foundational discoveries and advancements made by the founder led to the development of advanced gene therapy and AAV technology and over 500 patents for the same. AskBio’s state-of-the-art gene-therapy technology platform includes an industry-leading cell line manufacturing process, an extensive AAV capsid, and the promoter library. The company boasts of hundreds of proprietary third-generation AAV capsids and promoters capable of overcoming challenges associated with AAV vectors.
Bayer will have control over all of this and more. The company will have the full rights to AskBio’s gene therapy platform and the assets the platform churned out, including an internal pipeline of neuromuscular, central nervous system, and cardiovascular and metabolic gene therapies. Moreover, the company will acquire established contract development and manufacturing organizations that will be instrumental in laying the foundation for future opportunities in AAV gene therapies.
“As part of our strategy, we are building new therapeutic platforms including cell and gene therapies,” said Stefan Oelrich, Member of the Board of Management, Bayer AG, and President of the Bayer’s Pharmaceuticals Division. “As an emerging leader in the rapidly advancing field of gene therapies, the expertise and portfolio of AskBio supports us in establishing highly innovative treatment options for patients and further strengthens our portfolio. We want to help patients whose medical needs are not yet met by today’s treatment options, and we are looking forward to work together with the team at AskBio.”
While the deal’s intricate details are still under wraps, the company has decided to make an upfront payment of $2 billion to AskBio and potential success-based milestone payments of the same amount, making a total of $4 billion. Once the deal closes, AskBio will continue to operate as an independent company, a decision made to preserve the company’s entrepreneurial culture. Bayer has decided to bundle all its activities in the cell and gene therapy area under the newly established CGT unit to establish “an innovation ecosystem for the participating partners within the Bayer organization,” according to a statement.
Gene Therapy Market
In recent years, the gene therapy market has boomed due to fast technological advancements in the field. According to the USFDA, more than 200 applications of gene therapy drugs are expected by the end of this year. Scott Gottlieb, the former FDA commissioner, predicted that by the year 2025, the US would approve between 10 and 20 different gene therapies every year. This is in concurrence with a sudden increase in the number of companies developing gene therapies.
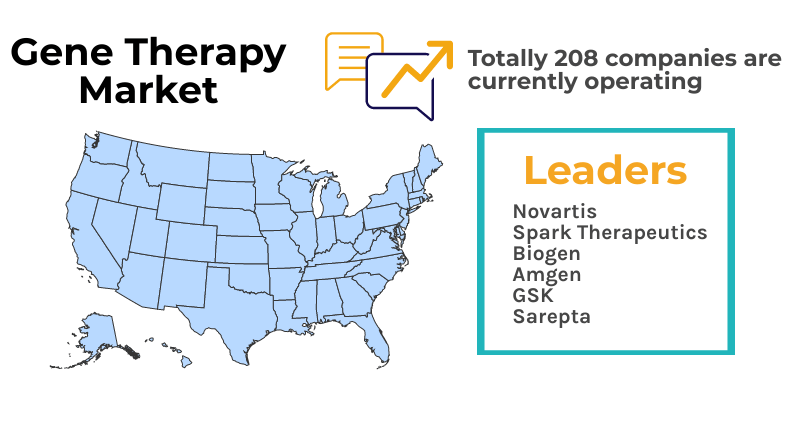
In North America, almost 208 companies are currently operating in this market, including leaders like Novartis, Spark Therapeutics, Biogen, Amgen, GSK, and Sarepta Therapeutics. Kymriah was the first ex-vivo gene therapy approved by the USFDA in 2017, followed by Yescarta in October the same year. Since then, two other therapies (Luxturna– Spark Therapeutics and Zolgensma- Novartis) in different indications have got the FDA’s blessing.
Looking at the unmet need to treat various genetic indications, many treatments are getting quickly approved with data from small trials. While it may seem that the FDA is cutting slack when evaluating these therapies, it recently delayed approval of BioMarin’s hemophilia therapy due to a lack of substantial data, leading to a potential setback for the sector.
Nevertheless, the companies are continuously making huge investments in the field, as clearly this is the future of therapies. For Bayer, it might be difficult initially to set footprints in the field where others are leading. It is also not clear if the latest acquisition will match the billions of dollars that the company is currently generating from drugs like Xarelto or Eylea. However, Bayer believes that this is a huge opportunity, and if therapies addressing heart failure and Parkinson’s are approved, they can make an incredible impact on the patients.
Related Article: Harvard Spinout Bags $1.8 Billion Worth Gene Therapy Deal with Roche
References
©www.geneonline.com All rights reserved. Collaborate with us: service@geneonlineasia.com









