Pharmaceutical Giants Expand Research and Manufacturing Capabilities, Fueling 2024 Innovation and Growth
In recent developments within the pharmaceutical and biotechnology sectors, various companies have announced significant investments, strategic shifts, and expansions that underline their commitment to innovation, research and development (R&D), and global health challenges. This comprehensive overview provides insights into these key movements, emphasizing their implications for the industry, workforce, and healthcare landscape.
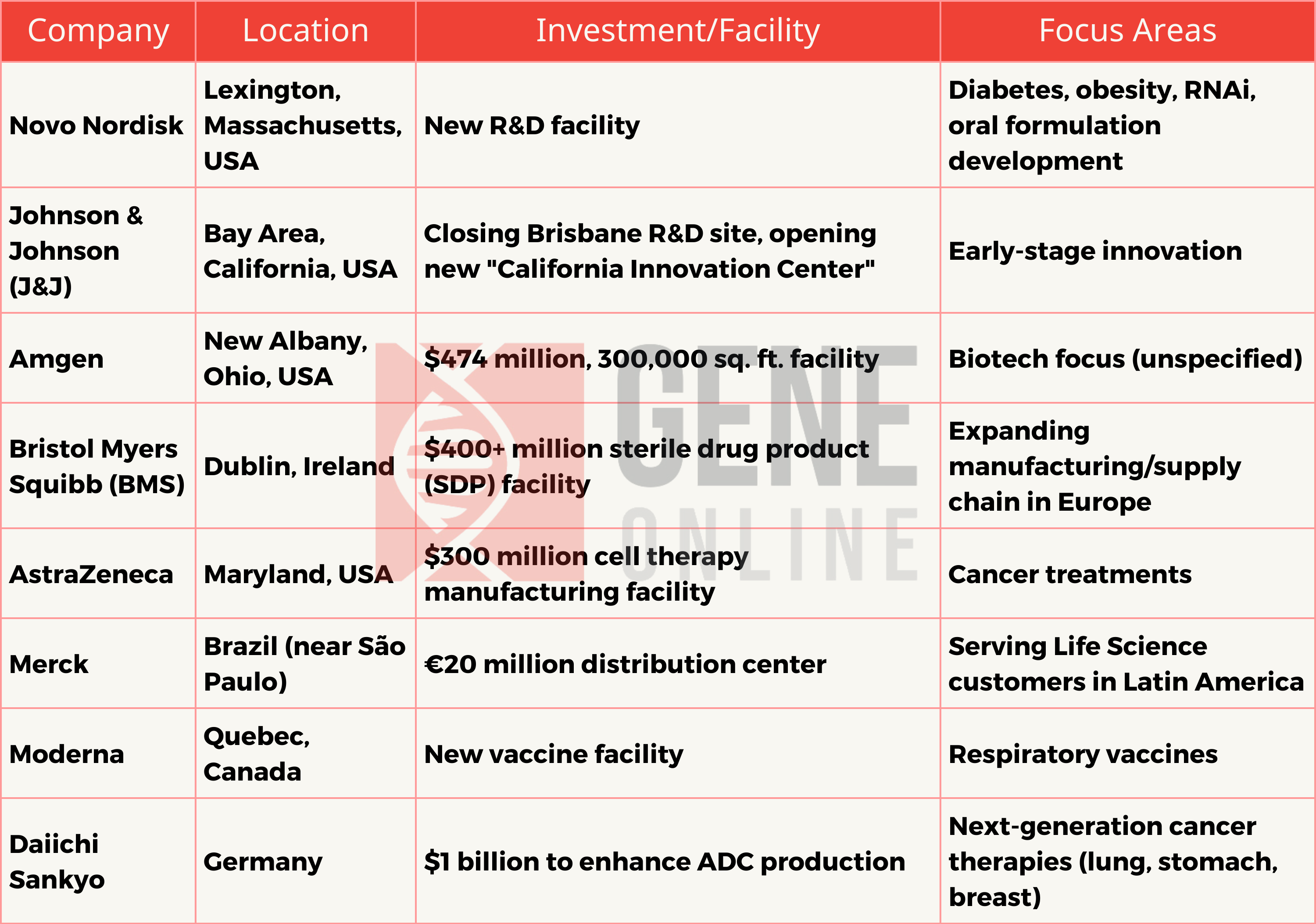
Novo Nordisk Enhances R&D Presence in Greater Boston
On February 28, Novo Nordisk, the Danish pharmaceutical behemoth, inaugurated a new R&D center in Lexington as part of its strategic consolidation of the U.S. R&D operations in the Greater Boston area. This expansion not only signals the company’s commitment to research and innovation but also underscores its intention to play a pivotal role in the U.S. pharmaceutical research landscape.
With plans to hire new staff in 2024, this move promises to bolster job opportunities in the region. Furthermore, Novo Nordisk is focusing on discovering breakthrough drugs beyond its blockbuster GLP-1 medications, Ozempic and Wegovy, marking a significant step toward its future endeavors in the pharmaceutical domain.
Johnson & Johnson’s Strategic Shift in California
On February 26, Johnson & Johnson (J&J) announced the closure of its R&D facility in Brisbane, California, operational for less than two years, instead, the company intends to open a new “California Innovation Center” in the Bay Area.
This strategic decision reflects a reorganization within J&J’s R&D operations, aiming to maintain an active presence in the early innovation ecosystem of the region.
While the closure may have local economic implications, the innovation center is anticipated to contribute to the Bay Area’s biopharmaceutical and technological landscape.
Amgen’s New Facility in Ohio
On February 26, Amgen completed a $474 million facility in New Albany, Ohio, which is expected to employ 400 full-time staff. This facility is not just a significant investment in the region but also a testament to Amgen’s commitment to innovation and growth in the biotech industry.
The collaboration with Columbus State Community College for a manufacturing apprenticeship program further highlights Amgen’s dedication to workforce development and skill-building initiatives.
Bristol Myers Squibb’s Acquisitions and Expansions
On February 26, Bristol Myers Squibb (BMS) is establishing a Science, Technology, and Innovation Centre in Hyderabad, India, with an investment of $100 million. The center aims to expand BMS’s drug development, IT, and digital innovation capabilities.
It is expected to employ 1,500 people and represents BMS’s first such center in India. This initiative highlights Hyderabad’s role as a life sciences hub and diversifies BMS’ global operations.
In addition, On February 22, Bristol Myers Squibb is investing over $400 million in building a sterile drug product facility.
AstraZeneca’s Bolster on Cell Therapy
On February 13, AstraZeneca is investing $300 million in a new manufacturing facility in Rockville, Maryland, to bolster its presence in cell therapy, focusing on cancer treatments. The facility is expected to be fully operational by 2026 and will create approximately 150 new jobs.
Additionally, the acquisition of Gracell Biotechnologies for $1.2 billion enhances its cell therapy pipeline, indicating a significant step towards launching its cell therapy platforms in the U.S. and advancing critical cancer trials.
Merck KGaA’s Expansion in Brazil
On February 28, Merck KGaA is inaugurating a new distribution center in Cajamar, São Paulo, Brazil, with an investment of $21.7 million (€20 million). This facility, covering 13,000 square meters, marks a significant upscale from its Cotia site, showcasing a considerable expansion of Merck’s operational capabilities.
Its strategic location near São Paulo, a hub for 60% of the country’s Life Science demand, aims to shorten delivery times, potentially to under 24 hours, enhancing service to local customers.
The facility includes a mezzanine, laboratories, administrative areas, and spaces for employee and customer amenities. In alignment with Merck’s global sustainability goals, the site has earned LEED Gold certification for its environmental and energy efficiency standards.
Moderna’s Vaccine Facility in Quebec
On February 27, Moderna established its first mRNA vaccine manufacturing facility outside the U.S. in Laval, Quebec, within the Biotech City area. Set to be operational by the end of 2024, this facility aims to produce vaccines against COVID-19 and other respiratory viruses, including influenza and RSV.
This development highlights Canada’s initiative to enhance domestic biomanufacturing and vaccine research capabilities. The Quebec region’s strong life sciences sector, bolstered by over 46,000 professionals and nearly 40,000 STEM students, positions Montreal as a key player in biopharmaceutical innovation.
Daiichi Sankyo’s Investment in ADC Production
On February 15, Daiichi Sankyo is investing around $1 billion in its Pfaffenhofen, Germany, facility to boost its antibody-drug conjugate (ADC) production, aiming to make it a global innovation center for ADCs. The expansion, expected to be complete by 2026, will create over 350 jobs by 2030.
This move is part of the company’s strategy to enhance its cancer therapy capabilities and maintain its leadership in the ADC market, with ADC sales projected to exceed $10 billion by 2029. The Pfaffenhofen site, traditionally focused on cardiovascular medicines, will now also prioritize oncological therapies, reflecting a significant contribution to the growing demand for ADC cancer treatments.
These strategic moves by leading pharmaceutical and biotech companies signify a robust commitment to advancing healthcare, research, and development across the globe. By investing in new facilities, expanding R&D operations, and pursuing strategic acquisitions, these firms are not only contributing to global health innovations but also creating new opportunities in the biopharmaceutical industry, reinforcing their roles as key players in the quest for groundbreaking treatments and therapies.
References:
- https://www.bizjournals.com/boston/news/2024/02/28/novo-nordisk-lexington-site.html?ana=RSS&s=article_search
- https://endpts.com/novo-plans-for-life-after-wegovy-setting-up-roots-in-boston-to-try-and-find-its-future/
- https://www.novonordisk.com/news-and-media/news-and-ir-materials.html
- https://endpts.com/jj-close-california-rd-facility-with-plans-to-open-new-innovation-center/
- https://www.bisnow.com/san-francisco/news/life-sciences/jj-to-close-research-bay-area-research-site-123072
- https://www.jnj.com/media-center
- https://www.beckershospitalreview.com/disruptors/amgen-builds-474m-facility-in-ohio.html
- https://www.amgen.com/newsroom/press-releases/2024/02/amgen-opens-state-of-the-art-biomanufacturing-site-in-central-ohio
- https://www.ndtvprofit.com/business/bristol-myers-squibb-inaugurates-100-million-it-drug-development-facility-in-hyderabad
- https://www.biopharma-reporter.com/Article/2024/02/22/bristol-myers-squibb-invests-over-400m-in-sterile-drug-product-facility?utm_source=RSS_Feed&utm_medium=RSS&utm_campaign=RSS#
- https://www.bms.com/media/press-releases.html
- https://www.constructiondive.com/news/astrazeneca-plant-rockville-maryland/707349/
- https://www.astrazeneca.com/media-centre/press-releases.html
- https://www.pharmaceutical-business-review.com/news/merck-distribution-centre-brazil/
- https://www.merckgroup.com/en/news.html
- https://www.costar.com/article/2020929266/moderna-completes-new-vaccine-facility-in-quebec
- https://www.modernatx.com/en-AU/newsroom-press-release
- https://www.pharmalive.com/daiichi-sankyo-drops-1b-to-boost-adc-manufacturing-capacity-at-german-site/?__cf_chl_tk=LybjLogktCjqvJeWe46XK.Mb2M1FAJkPewMZQXMovTg-1709189782-0.0-1469
- https://www.daiichi-sankyo.eu/media/european-news/news-detail/daiichi-sankyo-investiert-ca-eine-milliarde-euro-in-deutschland-standort-pfaffenhofen-bayern-wird-zu-internationalem-innovationszentrum-ausgebaut/
©www.geneonline.com All rights reserved. Collaborate with us: service@geneonlineasia.com









