BIO Asia Taiwan Review: The AI Tide, How Informatics Shape the Future of Pharmaceutical Industry
The continuous development of new drugs indicates the pharmaceutical industry is still going strong. As technology continues to reinvent itself with time, various IT tech are introduced to support drug development. Unfortunately, the Eroom’s Law still applies to the current development status, drug discovery becomes more difficult and more expensive despite the advancement of science. However, in recent years the overall elevation of computational capacity had boosted the application of big data and artificial intelligence in drug development, bringing high anticipation to improve innovation efficiency.
In this year’s BIO Asia Taiwan Exhibition, many companies demonstrated how to utilize big data analysis and AI technology in product development and problem solution discovery (Pic 1.), we have picked the highlights for this events:
CYTLIMIC
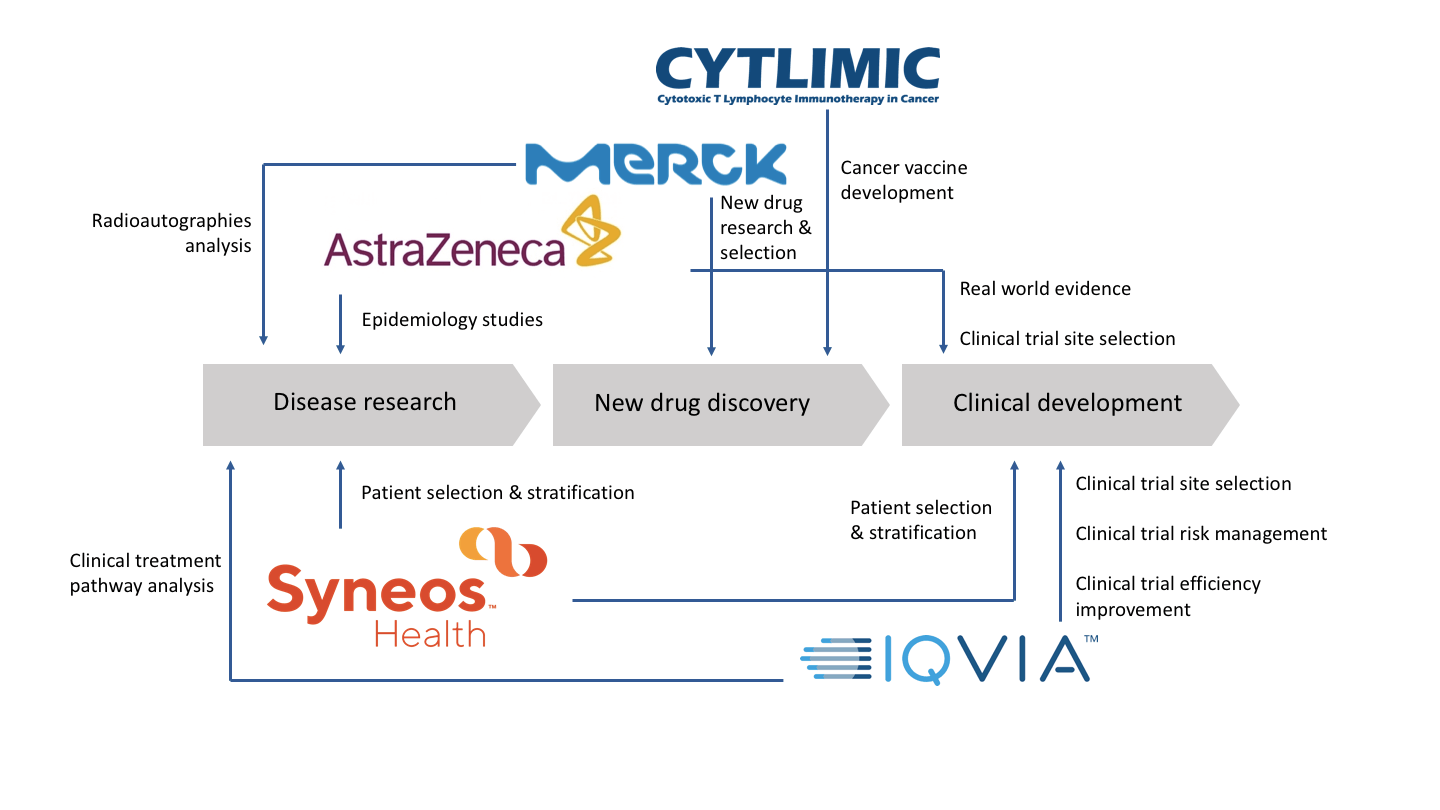
CYTLIMIC (NEC) is one of the pioneers of utilizing AI in medical development. Before they split from NEC, they were already predicting immune response via machine learning back in 2001. They analyzed the sequence of antigen peptides and predicted affinity between antigen peptides and human leukocyte antigens (HLA).
CYTLIMIC discovered the therapeutic cancer vaccine CYT001 with the support of AI technology, it includes 2 antigen peptides (HSP70 and GPC30) and adjuvant (Poly-ICLC and LAG-3Ig). Currently, one clinical trial is completed and two are still ongoing (YNP01, YCP02, and CRESENT1). The target indications are for hepatocarcinoma and gastric carcinomas. Through analysis of pathological samples from the initial trial, it is discovered that CYT001 can increase both the infiltration of toxic T cells in the tumor, and also the number of peptide-labeled cancer cells recognized by the toxic T cells, allowing the cold tumors transform into hot tumors that are more reactive to the immune system.
AstraZeneca
As one of the major players in the medical industry, AstraZeneca aggressively introduces big data and AI technology to improve drug development: (1) To use propensity score matching on real world data to find comparable control groups for single-arm clinical trials. (2) To use PageRank algorithm to analyze how patients search for recommended doctors, and further support trial site selection. (3) AstraZeneca cooperated with three other facilities, including the Public Health England (PHE) to create a simulation model of National Cancer Registration and Analysis Service (NCRAS) via machine learning. It allows other parties to perform analysis on the simulation model, gain certain parameters and provide the codes to the analysts of NCRAS to register them into the actual system to obtain final results.
Merck KGaA
Merck’s KGaA is already cooperating with AI drug development companies, CYCLICA and IKTOS, to enhance the efficiency of drug development. AI has been used to build pharma kinetic models (ADMET), set up quantitative structure-activity relationship models, docking, and support in silico drug candidate selection and predict possible pharmacological, adverse effects and toxicity.
Besides, Merck KGaA also uses textural analysis on colorectal cancer radioautographies, combining biolabeling protein to build prediction models of how patients may react to the drugs.
IQVIA
In this BIOAsia, the two CRO giants IQVIA and Syneos Health had demonstrated the introduction of big data and AI technology to solve difficult problems. For IQVIA, it includes (1) Integrating experience of past clinical trials, project leaders and trial location specificities to build an AI model to assist in trial locations selection, provide risk management and accelerate trial initiation. (2) Adding a time parameter to the clinical trial data, and create treatment pathways based on this information. It assists sponsors to discover new opportunities or to design future trials. (3) Adding NLP to non structural data like medical records, this can enrich data, and establish risk prediction models to assess the quality of the clinical trial.
Syneos Health
Syneos Health combines genomics, proteomics, metabolomics, and clinical data together to categorize patients with supervised or unsupervised learning AI technology. Gene ontology is then further applied to assist clients to further patients selection and stratification.
Syneos Health takes a step further to categorize patients that don’t respond to trial therapy. Although it is the application of post-hoc analysis or retrospective studies, but it can still solve some uncertainties, and provide clinical evidence or support the hypothesis design for the following trial.
During this event, we can see that many companies have actively involve AI technology to support new drug development. Though many challenges such as unstandardized data interpretation, data accessibility, data rights and privacy challenges are still presented. However, we can expect that the usage of data-driven model in product development and strategy will dominate and reshape the pharmaceutical industry. These technology don’t just pursue the success of single respective product, but aim to elevate the competitiveness of production pipeline as a whole, provide a more comprehensive approach of drug research, and elevate the efficiency to deliver new drugs to the market.
©www.geneonline.com All rights reserved. Collaborate with us: service@geneonlineasia.com









